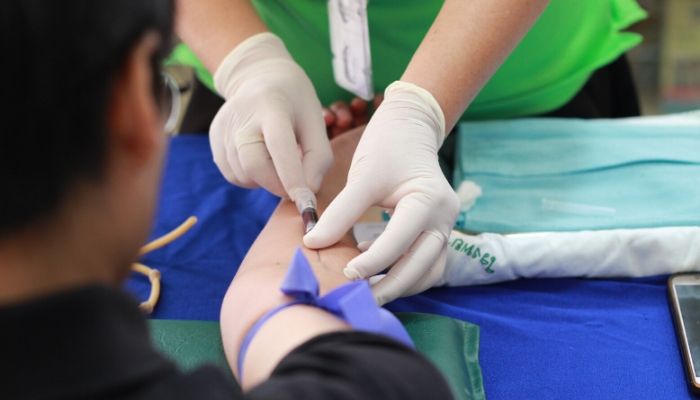
The government announced that it has begun a large-scale test study of COVID-19 in the UK’s general population. The major study will test up to 300,000 people in their own households over a twelve-month period. It will also include antibody testing to help understand levels of immunity as well as the current rate of infection.
Led by the Department for Health and Social Care (DHSC) and the Office for National Statistics (ONS), the study forms part of Pillar 4 of the government’s COVID-19 testing strategy. The results will help scientists and the government in the ongoing response to the coronavirus outbreak including the development of new treatments, with initial findings expected in early May.
Participants in the study will form a representative sample of the UK population by age and geography. They will provide swab samples and answer questions during a short home visit from a trained health worker, before being monitored at various intervals throughout the year. Adults from around 1,000 households will also provide a blood sample to help determine what proportion of the population has developed antibodies to COVID-19.
National Statistician Sir Ian Diamond said: “The Office for National Statistics has huge experience in running very large household surveys that gather vital information from a genuinely representative sample of the entire population. In this case we’ll be using that capability to help our health expert colleagues to create a reliable picture of the scale of COVID-19 infection and antibody development that will inform the key decisions that lie ahead in this pandemic.”
The study brings together scientific expertise from a number of world-leading organisations, including study sponsor University of Oxford, data science company IQVIA UK and the National Biosample Centre in Milton Keynes. It’s hoped that, alongside testing in other countries such as the US and Germany, the study will provide global data that will transform our understanding of the virus. Large-scale antibody testing could allow more individuals to safely return to work, while also contributing to the eventual development of a possible vaccine.
On the 23 April, the Medicines and Healthcare products Regulatory Agency (MHRA) approved the COVID-19 Oxford Vaccine Trial request to trial a vaccine in a little over one working week. The application was made on 18 March, and on 26 March the MHRA gave the COVID-19 Oxford Vaccine Trial the green light. Scientists in Oxford are ready now to start the first clinical testing phase to determine whether the vaccine is safe and effective.
ContractingWISE has a range of options to help you keep your contracting career on track, including setting up or reopening a limited company, or finding an umbrella company to suit your needs. To talk to a member of our team, call: 0203 642 8679



